VACCELERATE Clinical Studies
Clinical trials serve the purpose of clarifying open questions regarding new vaccines, therapies or medicines. By participating in clinical trials, you can make a major contribution to scientific progress. As a study participant you support the research of diseases and the development of new treatment options and at the same time receive comprehensive medical care.
VACCELERATE is carrying out three COVID-19 vaccine trials. The results will be linked here as soon as they are published.
STUDY 1: EU-COVAT-1 AGED



A Multinational, Phase 2, Randomised, Adaptive Protocol to Evaluate Immunogenicity and Reactogenicity of Different COVID-19 Vaccines Administration in Older Adults (≥75) Already Vaccinated Against SARS-CoV-2
Lead: Oliver Cornely, Institution/Sponsor: UHC, Germany
Details:
https://clinicaltrials.gov/ct2/show/NCT05160766?cond=Covat+aged&draw=2&rank=1
Publications:
STUDY 2: EU-COVAT-2 BOOSTAVAC


An International Multicentre, Phase 2, Randomised, Adaptive Protocol to determine the need for, optimal timing of and immunogenicity of administering a booster mRNA vaccination dose against SARS-CoV-2 in the general population (18+ years) already vaccinated against SARS-CoV-2
Lead: Patrick Mallon, Institution/Sponsor: NUID UCD, Ireland
EudraCT-number: 2021-004889-35
Details:
https://twitter.com/vaccelerate_eu/status/1503688207506063360
https://www.clinicaltrialsregister.eu/ctr-search/trial/2021-004889-35/IE
STUDY 3: EU-COVPT-1 COVACC



A Phase 2, Comparative Randomised Trial to Evaluate the Impact of Reduced COVID‐19 mRNA Vaccination Regimen on Immunological Responses and Reactogenicity in Paediatric Subjects with Prior SARS‐CoV‐2 Immunity
Lead: Patricia Bruijning-Verhagen, Institution/Sponsor: UMCU, Netherlands
EudraCT number: 2021-005043-71


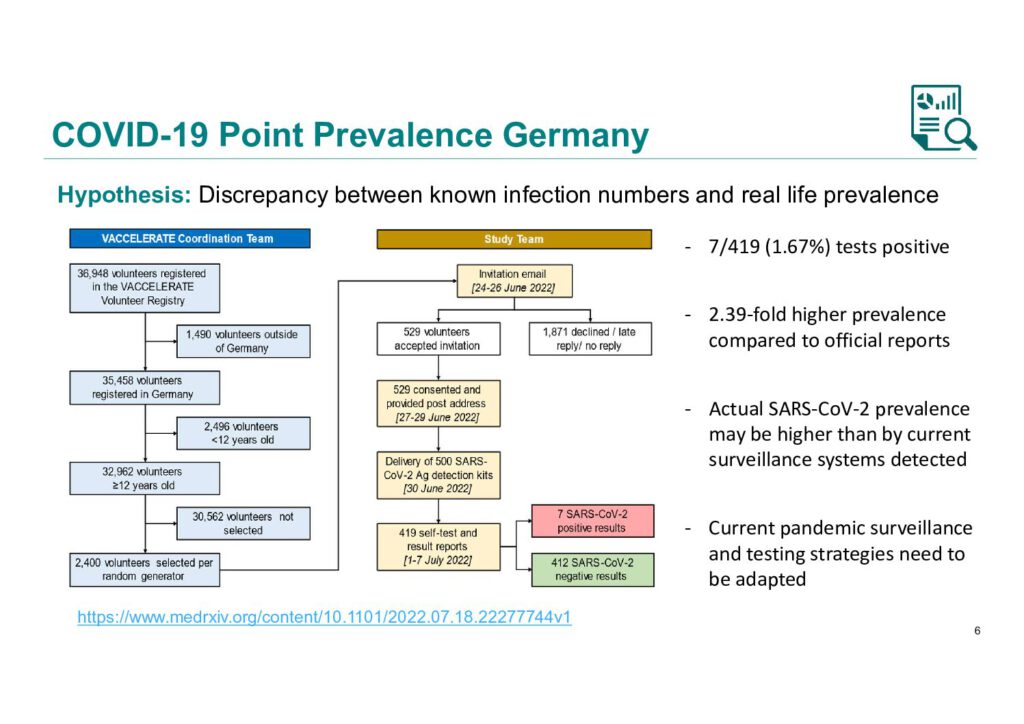
COVID-19 Point Prevalence Germany

VACCELERATE Mpox Initiatives
All information on the VACCELERATE mpox initiatives including the mpox standard, ooperations and publications can be found here: https://vaccelerate.eu/mpox/
Press Releases and Articles on the VACCELERATE Clinical Studies


Publication Video: https://www.youtube.com/watch?v=xTTTNcENoSc


